A Novel AI Approach Utilizing Large Language Models and GWAS to Validate Drug Target Efficacy and Predict Adverse Effects
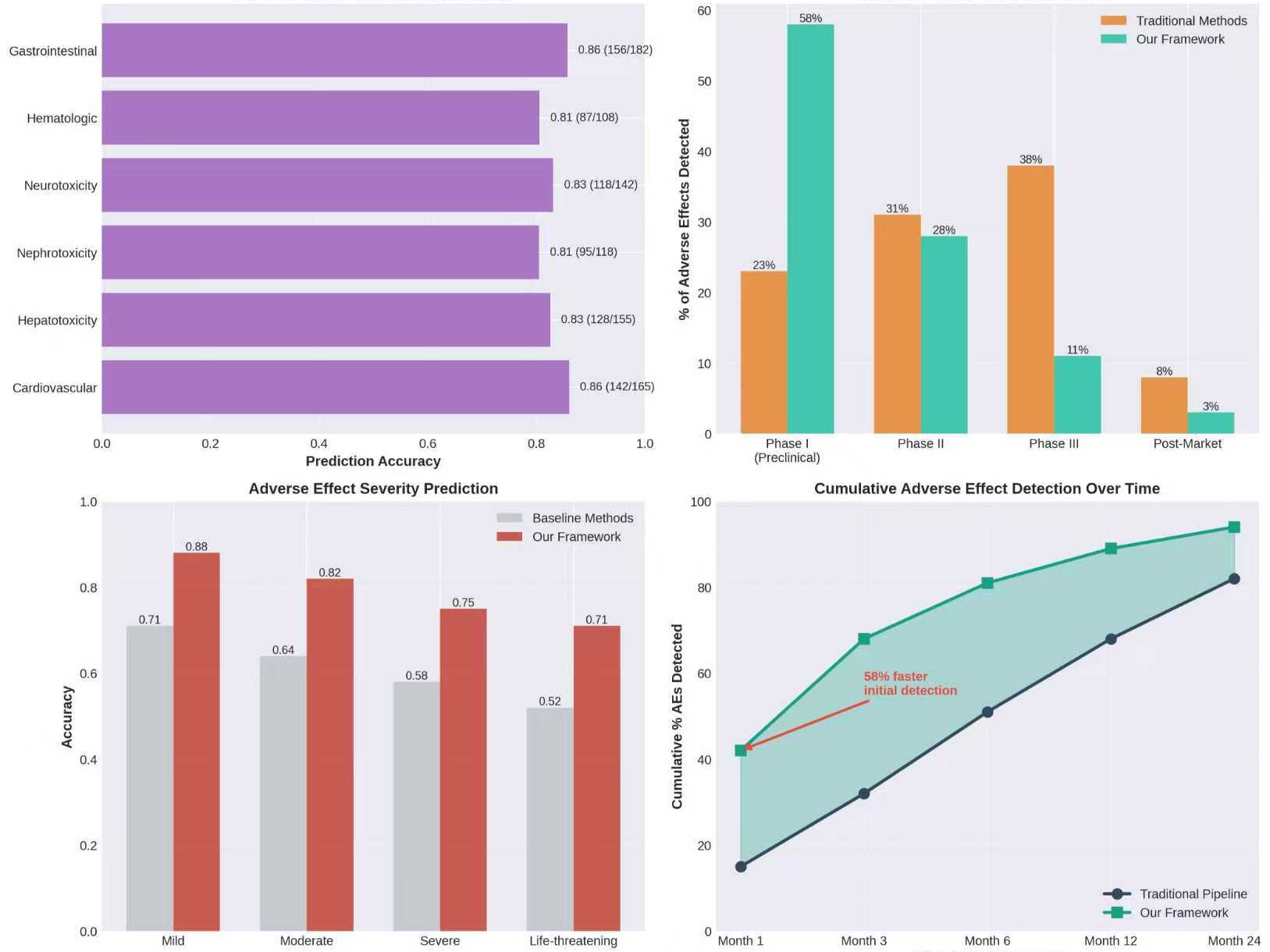
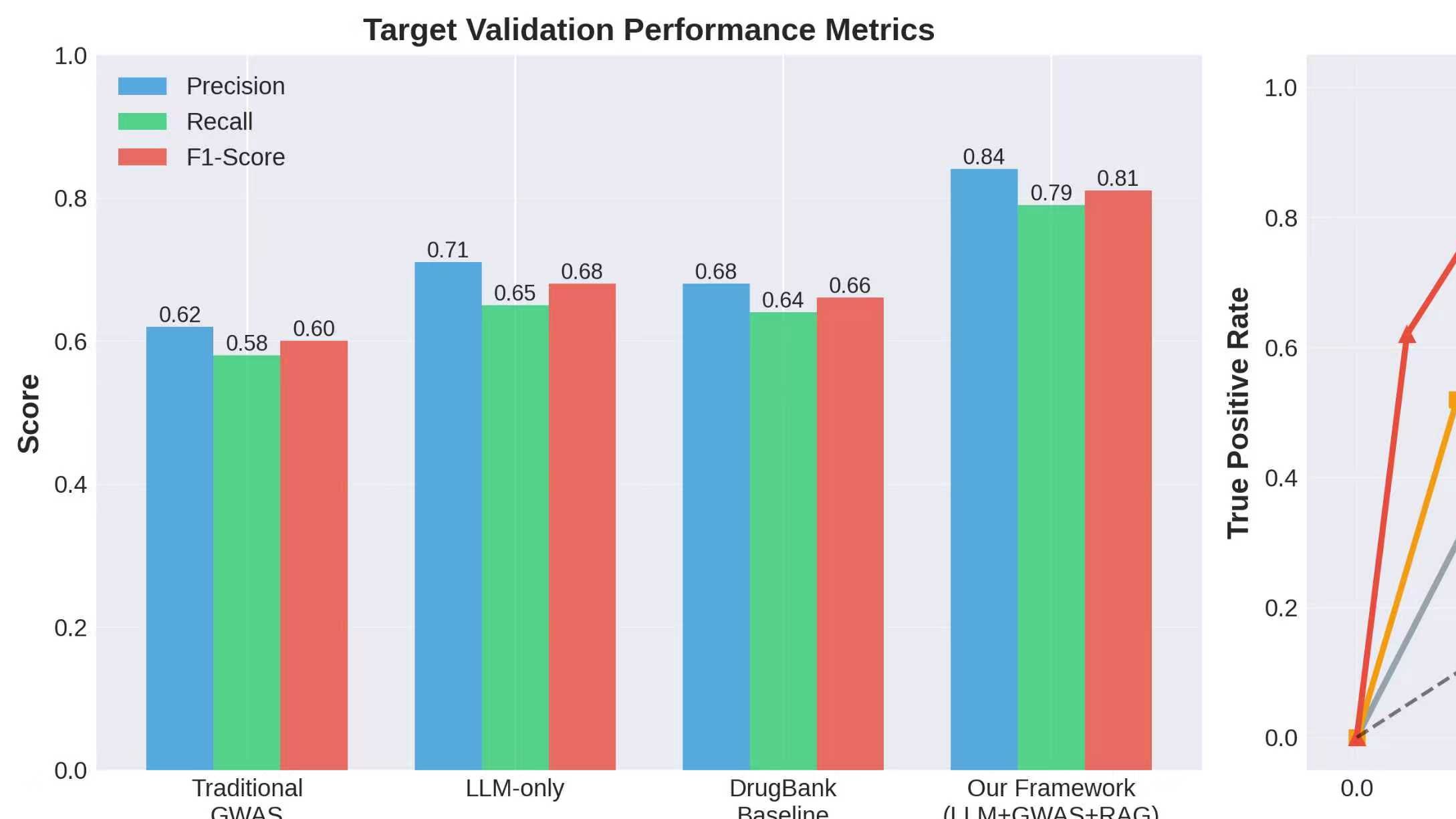
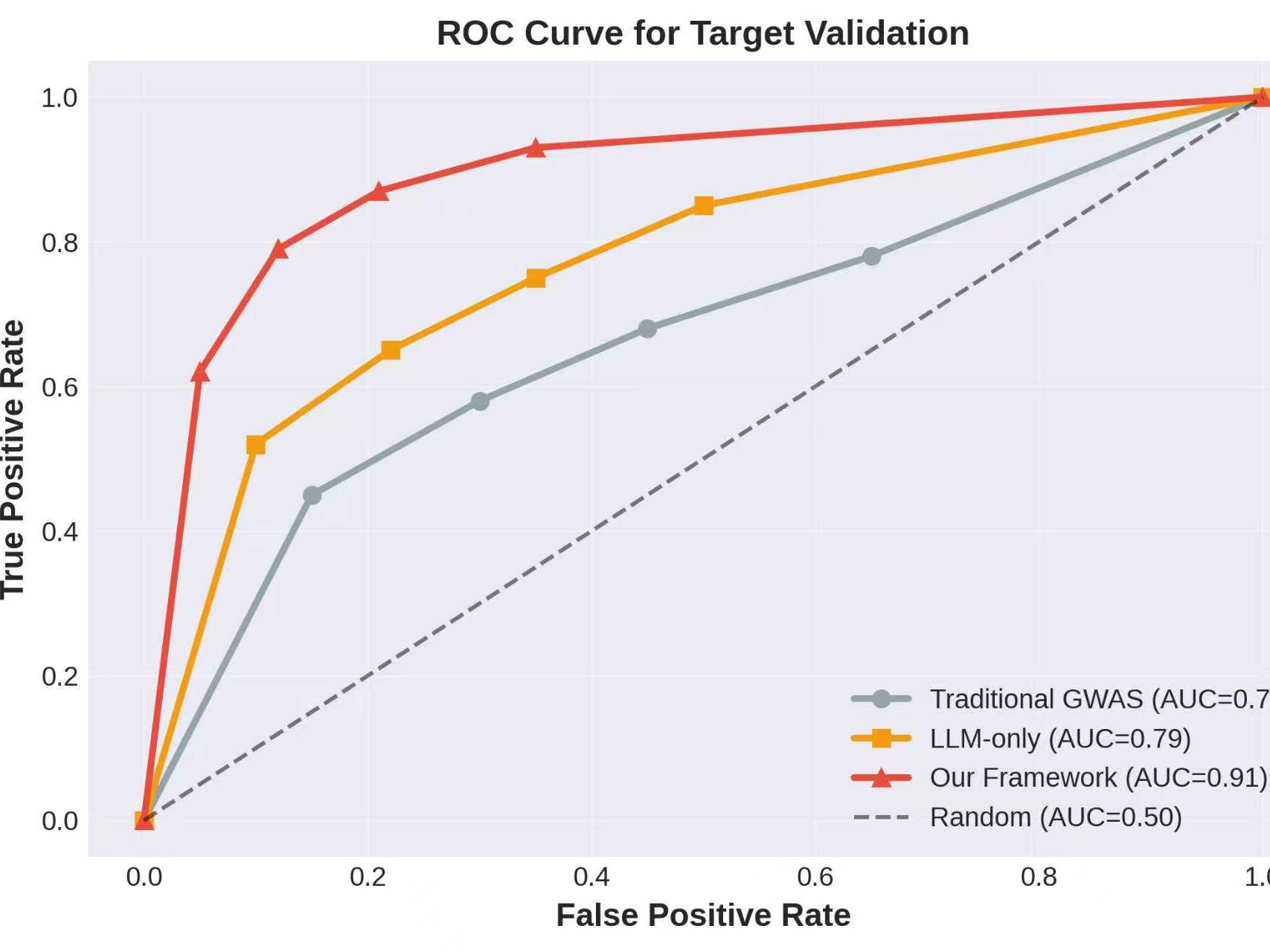
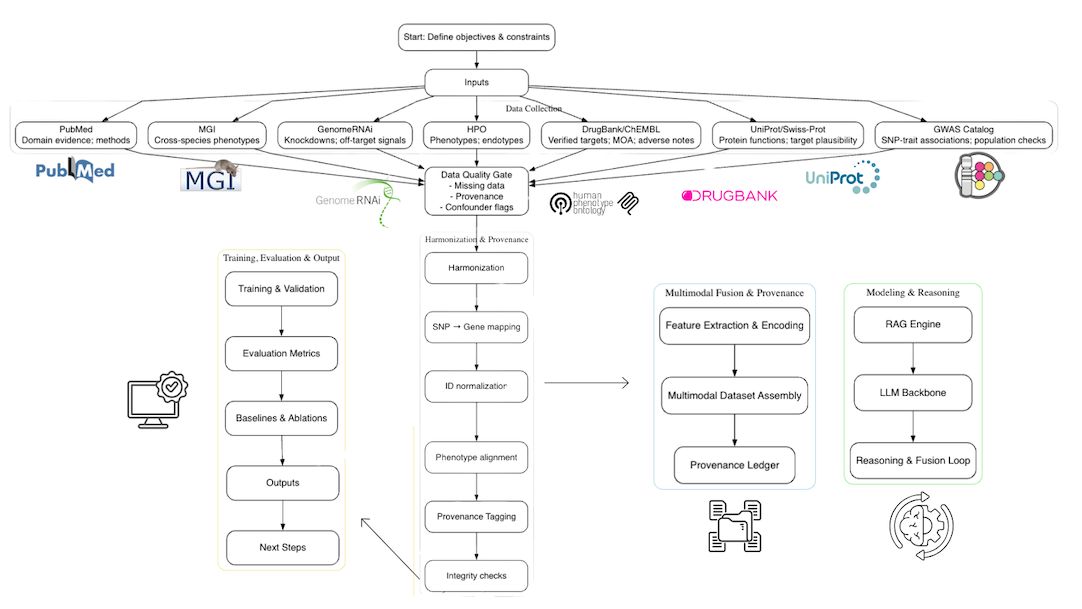
The early-stage failure of drug candidates remains a significant challenge in pharmaceutical development, primarily driven by inadequate validation of drug targets and unforeseen adverse drug reactions. This study introduces a novel framework that integrates Large Language Model (LLM)-driven reasoning with Genome-Wide Association Study (GWAS) data to improve drug target validation and predict adverse effects in the early phases of drug development. The integrated framework aggregates data from various biomedical sources, including the GWAS Catalog, UniProt, DrugBank, and others, to enhance target prioritization and safety analysis. Evaluations revealed that the system outperformed traditional methods, achieving an Area Under the Curve (AUC) of 0.91 in target validation and improving adverse effect prediction accuracy by 24%. Specifically, the framework detected 58% of potential adverse effects during Phase I/preclinical stages, substantially higher than the 23% detected by traditional methods. These results highlight the potential of AI-enhanced drug development processes to reduce costs and accelerate timelines.